

We are focusing on the differentiation and migration stages of cortical development. Neurons are born in the ventricular zone (germinal zone, neuroepithelium) and migrate radially and/or horizontally to form the neocortex. We have newly cloned several novel genes that are expressed in the neuroepithelium and have elucidated the functions of these molecules.
We cloned a novel gene (which we call 'FILIP') the mRNAs of which are located
in the ventricular zone. FILIP regulates the movement of cortical neurons
out of the ventricular zone by inducing the degradation of an actin-binding
protein, filamin A (Nagano, T. et al., Nature Cell Biology, 4: 495-501, 2002). When to start migration is essential to the formation
of the 6 layers of the neocortex, which are a typical feature of this part
of the brain. FILIP is a key molecule controlling this process. The outline
of our work is introduced in the highlight section of 'Nature Reviews Molecular Cell Biology, 3:472-473. 2002'. Later, we reported that the amount of filamin A is crucial for the neuronal
shape of radially migrating neurons (Nagano, T. et al., Journal of Neuroscience, 24(43):9648-9657, 2004. This paper was selected as a highlight paper). We are now studying
FILIP knockout mice and have found that FILIP is involved in neuronal transmission
(Yagi et al., Scientific Reports, 4, 6353; DOI:10.1038/srep06353). Concurrently, we also elucidated one
of the fundamental mechanisms of directed cell migration and cortical development
(Takabayashi et al., Journal of
Biological Chemistry, 285(21):16155-16165, 2010; Xie et al., Cerebral Cortex, 23(6):1410-1423, 2013).
In addition to the works mentioned above, we have carried out a couple of collaborative works to address further the mechanisms underlying the cortical development. In conjunction with Horio’s lab at Sapporo Medical University, we elucidated the significance of SIRT1 (Hisahara et al., Proc. Natl. Acad. Sci. (USA). 105 (40):15599-15604, 2008, which was selected as the cover for this issue and was introduced in the “cutting edge” section of Cell) in neuronal differentiation. Working with Hirotsune’s lab at Osaka City University, we found that modulating calpain activity can be a potential therapeutic approach for lissencephaly due to Lis1 mutation (Yamada et al., Nature Medicine, 15(10): 1202-1207. 2009).
The neocortex is well-developed in mammals, especially in primates. It
serves as the center for higher cerebral functions such as integration
of multi-modal sensory inputs and regulation of voluntary motions. It consists
of many functional areas such as visual, auditory, motor, and association
areas, each of which processes information of a certain modality or property.
Inter-areal connections underlie these higher functions, comparing and
integrating information processed in each area. Moreover, these areas are
not interconnected at random. Hierarchical organization of such subdivisions
are crucial for the neocortex to exert its higher cognitive functions.
It has been revealed that the disorganization of such hierarchy is involved
in certain psychiatric diseases and developmental disorders. However, how
hierarchical organization is formed and works have remained fully open
for a century.
(1) Developmental mechanisms of
the inter-areal connections in the cerebral neocortex.
We are analyzing the developmental mechanisms of these neural circuits
using the mouse brain. We have already developed a way to manipulate some
fundamental connections specifically. With our newly developed technologies,
we are currently studying how such connections are formed and work.
(2) Mechanisms of subtype-specific circuit formation of neocortical neurons.
The neocortex is made up of many different types of neurons in terms of
connections, e.g. inter-areal, local, inter-hemispheric, and subcortical
connections. These neurons are located in specific layers of the neocortex,
often intermingling with each other. How these neurons with different connection
patterns are fate-determined and project their axons properly according
to their types during development is a largely unanswerd question. We are
analyzing the mechanisms of neuronal subtype specification and respective
circuit formation by comparing neurons having inter-areal connections with
those having inter-hemispheric ones.
The neocortex controls many subcortical regions. Especially, what we call
the ‘pyramidal tract’ is one of the major subcortical projections and is
essential for exerting motor function. During the development of the pyramidal
tract, axon collaterals protrude from their main shaft toward nuclei such
as the superior colliculi, pontine nuclei, and inferior olivary nuclei.
However, the molecular mechanisms by which axon collaterals are induced
and project to specific regions of the brain are mostly unknown.
We are currently studying how the pyramidal tract is formed, especially
focusing on how collaterals from the main pyramidal tract target the pons,
which is the most important relay structure between the cerebral cortex
and the cerebellum. We have developed gene expression/ knockdown techniques
including the in utero electroporation method, primary culture of cortical
neurons, and organ culture of the brain tissues in collagen gels to examine
the pyramidal tract and/or pontine nuclei. Using these techniques we are
now trying to identify the molecules involved in the regulation of collateral
formation and to disclose the signaling mechanisms that reorganize the
cytoskeleton during this axon collateralization.
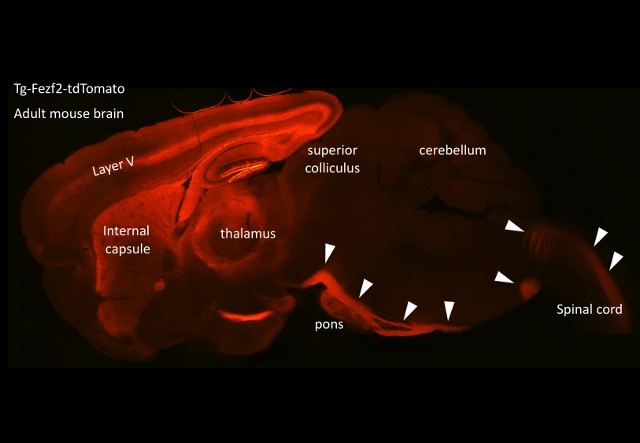 Although we are focusing on this pyramidal tract projection, underlying
molecular mechanisms are so common that we can apply our knowledge to find
a way to rebuild or repair the brain as well as to understand/cure other
neurological diseases caused by malformation/malfunction of neural circuits.
Although we are focusing on this pyramidal tract projection, underlying
molecular mechanisms are so common that we can apply our knowledge to find
a way to rebuild or repair the brain as well as to understand/cure other
neurological diseases caused by malformation/malfunction of neural circuits.
The incidence of developmental disorders has increased rapidly, and social
demands to treat such disorders are growing. They affect close to, or more
than, 1 % of all children worldwide. Concurrently, it has been recognized
that there exists a wide variation in symptoms in developmental disorders
such as autism. Such variations have led to the concept of autism spectrum
disorder (ASD). Many responsible mutations in the genome for autism spectrum
disorders have been reported, but how they result in similar behavioral
patterns has remained an open question. Moreover, the development of curative
medicines has not been successful. Recently we made an original mouse model
for studying ASD. We are currently tackling this issue with our model
A salient feature of neuronal cells is that they are segmented with spatially
and functionally isolated microdomains, such as myriads of synapses and
growth cones, that are distant from the cell body, which is the sole center
for gene expression from the genome. Accordingly, such spatial sequestration
must make differences in qualitative and quantitative requirements for
proteins among the microdomains, depending on which microenvironment they
face. Since these proteins would supposedly be synthesized in the cell
body and recruited into each of the targeted microdomains by a protein-transport
system, it has become a complicated task to explain the mechanism that
enables the delivery of the required proteins to the required domains in
response to external stimuli. However, the discovery of localized mRNA
molecules in dendrites and growth cones has solved this problem, in part;
i.e., on-demand and in situ protein synthesis from these mRNAs transported
to the microdomains in advance may allow such delivery. Especially, a group
of resident mRNAs in the dendrites (dendritic mRNA), such as αCaMKII, β-actin,
and the mRNAs of certain glutamate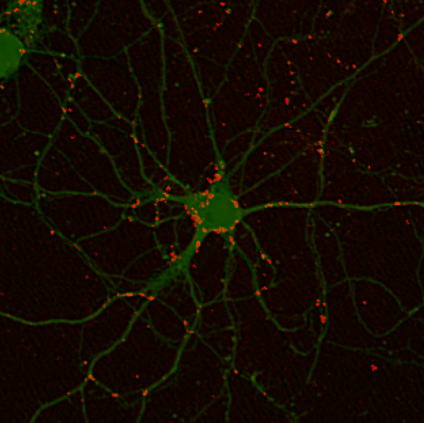 receptors, have been reported to be translated in the postsynaptic regions.
Thus, the neighboring translation to the regions where the products can
work would overcome spatiotemporal complexities that accompany the one-by-one
protein transport model. Among dendritic mRNAs, we are focusing on αCaMKII
mRNA, the product of which is a key regulator of synaptic transmission
and shape, and have identified cis-element regions for dendritic targeting
and a list of proteins that bind to the 3’-untranslated region, including
the cis-element. Further studies on the functions of these identified proteins
for dendritic targeting are now in progress.
receptors, have been reported to be translated in the postsynaptic regions.
Thus, the neighboring translation to the regions where the products can
work would overcome spatiotemporal complexities that accompany the one-by-one
protein transport model. Among dendritic mRNAs, we are focusing on αCaMKII
mRNA, the product of which is a key regulator of synaptic transmission
and shape, and have identified cis-element regions for dendritic targeting
and a list of proteins that bind to the 3’-untranslated region, including
the cis-element. Further studies on the functions of these identified proteins
for dendritic targeting are now in progress.